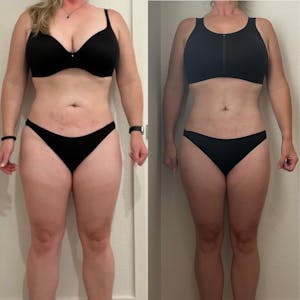
Tirzepatide is an injectable medication initially developed for the treatment of type 2 diabetes, but recent studies have indicated its potential for assisting in weight loss. This innovative treatment mimics the effects of naturally occurring hormones that regulate appetite and food intake. As a medication that affects metabolic regulation, Tirzepatide has shown promising results in improving blood sugar levels and helping individuals achieve significant weight loss.
An Innovative Approach to Managing Obesity
At North Shore® Cosmetic Surgery, we understand the challenges you might face on your journey to a healthier weight. Tirzepatide could be a powerful tool in your weight management arsenal when diet, exercise, and other medications have not yielded the results you desire. By incorporating this treatment into your health plan, you may find an effective path to shedding excess weight and moving towards your wellness goals.
How Does Tirzepatide Help Weight Loss?
Tirzepatide supports weight loss through its dual-action mechanism, which targets critical hormones in appetite regulation and glucose metabolism. It is designed to mimic and enhance the effects of incretins, which play a significant role in managing body weight.
GLP-1 Receptor Agonism
- Tirzepatide activates GLP-1 receptors, which, in turn, stimulates insulin secretion in response to high blood glucose levels
- By activating these receptors, Tirzepatide can decrease your appetite, leading to reduced calorie intake and subsequent weight loss
GIP Enhancement
- Tirzepatide enhances the action of GIP (glucose-dependent insulinotropic polypeptide), another incretin hormone.
- GIP amplifies the effect of GLP-1, directly reduces food intake, and increases energy expenditure, assisting you in weight management
Is Tirzepatide Safe for Weight Loss?
Tirzepatide, as a medication, has undergone clinical trials to evaluate its effectiveness and safety. The U.S. Food and Drug Administration (FDA) has a process to review and approve new medications based on this data.
Clinical trials for Tirzepatide have shown that it can lead to significant weight loss. However, these studies also monitor for side effects to ensure the benefits outweigh the risks.
Common side effects may include:
- Nausea
- Diarrhea
- Reduced appetite
While minor side effects are relatively common, severe reactions are less frequent. If you start Tirzepatide, your healthcare provider at NSCS will monitor your progress and check for any adverse effects during treatment.
It's important to note that as of the last known update before January 2024, Tirzepatide had been approved by the FDA for treating Type 2 diabetes. Weight loss may be a beneficial side effect, and while it shows promise, the status of its approval specifically for weight loss may be subject to change.
Am I a Good Candidate for Tirzepatide for Weight Loss?
When considering Tirzepatide for weight loss, it's essential to evaluate specific qualifications that may make you a suitable candidate.
- Existing Medical Conditions: Do you have Type 2 diabetes? Are you struggling with obesity or overweight with a related condition (e.g., high blood pressure)?
- Previous Weight Loss Efforts: Have you tried diet and exercise without achieving your desired weight loss?
- BMI Criteria: Candidates must have a body mass index (BMI) of 30 kg/m² or greater (obese). Alternatively, they may have a BMI of 27 kg/m² or greater (overweight) with at least one weight-related condition.

Lifestyle Considerations and Long-Term Management Strategies
When incorporating Tirzepatide for weight loss, it’s vital to integrate lifestyle modifications to enhance and sustain its benefits. Committing to a healthy lifestyle is the foundation for long-term weight management success.
- Diet: Focus on a balanced diet rich in whole foods and grains and lean proteins like poultry, fish, beans, lentils
- Exercise: Regular physical activity is crucial. Engage in at least 150 minutes of moderate-intensity aerobic activity weekly. Include muscle-strengthening activities on two or more days a week.
- Behavioral Changes: Adopt strategies to support your efforts. Engage in mindful eating and practice relaxation techniques such as meditation or deep breathing.
- Monitoring: Keep track of your progress by recording your weight and body measurements monthly. Use journals or apps to stay accountable regarding food and activity logs.
Adhering to these synergistic strategies isn't just about the short-term gains; it's about cultivating a sustainable and healthy life approach that complements Tirzepatide. By making these lifestyle adjustments, you work towards weight loss and improved overall health.
Why Choose North Shore® Cosmetic Surgery in Melville, NY, for Tirzepatide?
Our board-certified plastic surgeons in Long Island, including Dr. James Romanelli, Dr. John Layliev, and Dr. Yee Cheng Low, are recognized for their expertise and exceptional outcomes. Their individualized approach and advanced surgical techniques ensure your Tirzepatide treatment is tailored to your needs.
Your journey with us is not just about the procedure itself but encompasses comprehensive care from consultation through recovery. We aim to provide a supportive, welcoming environment, ensuring that you feel comfortable and informed every step of the way. With a focus on personalized care and high patient satisfaction, our center has received glowing reviews, showcasing our commitment to not just meet but exceed your expectations.
Choosing North Shore® Cosmetic Surgery for your Tirzepatide regimen means entrusting your care to a team that prioritizes your well-being and satisfaction in an environment that sets the bar for excellence in weight loss. Contact us today to schedule your consultation.